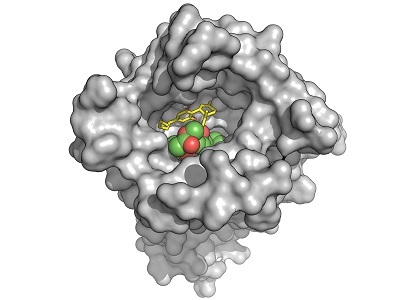
CBD (yellow stick structure) interferes with binding of an opioid (green and red) by stabilizing an opioid receptor (gray) in its inactive form. Credit: Charles Kuntz
In January, the Center for Forensic Science Research & Education (CFSRE) issued a public health alert that a new nitazene analog up to 20x more potent than fentanyl was spreading from Pennsylvania to Florida.
In that alert, the organization said, “Higher doses and/or redosing of naloxone may be needed based on clinical signs and symptoms, [but] there has been no evidence of any new naloxone-resistant synthetic opioids.”
While that’s good news on the surface, it’s not the first time CFSRE has had to recommend more than one dose of naloxone to reverse an overdose. In fact, as novel psychoactive substances get more potent, it’s becoming more and more common to administer multiple doses of naloxone.
Compared with other compounds such as heroin or morphine, fentanyl and its other synthetic relatives bind more tightly to opioid receptors in the brain. Naloxone reverses an overdose by competing with the drug molecules for the same binding sites on the receptors. But because fentanyl binds so readily, it has a leg up on naloxone.
That’s why researchers at Indiana University are investigating naloxone alternatives, or at least something that could work synergistically with naloxone to strengthen its effects.
In research presented at the American Chemical Society Spring 2023 meeting this week, the team says they found success in an unexpected place—another drug. The Indiana researchers are now looking to cannabidiol (CBD), a component of marijuana, as a possible alternative.
The team previously published work on CBD as a negative allosteric modulator at a separate opioid receptor, which is critical in bringing about the effects of cannabis.
“Allosteric modulators bind to a different site on a receptor so there's actually no direct competition with fentanyl,” explains graduate student Taryn Bosquez-Berger. “Once we realized that CBD was also a negative allosteric modulator—or could potentially be a negative allosteric modulator at opioid receptors—it wasn't a big logical leap.”
The research team then altered CBD’s structure to generate derivatives. Bosquez-Berger tested the new compounds in cells with a substance called DAMGO, an opioid used only in lab studies. She then monitored a molecular signal that diminishes when this type of drug binds. Armed with feedback from these experiments, the team further refined the structures.
In the end, they narrowed the field to 15, which they tested at varying concentrations against fentanyl— with and without naloxone.
They found that several derivatives reduced fentanyl binding even at what Bosquez-Berger described as “incredibly low” concentrations. Several also outperformed naloxone’s opioid-blocking performance. Two derivates showed a synergistic effect when combined with the antidote.
The team has since begun testing the most successful derivatives in preclinical mouse models to see if the compounds alter behaviors associated with taking fentanyl.
“We are looking at the ability of these compounds to reverse respiratory depression, because respiratory depression is what causes these fentanyl-induced deaths. Your skeletal muscles tense up and you can't breathe, so we're really interested in looking at how these compounds can reverse that effect,” said graduate student Jessica Gudorf, who is also working on the project.
Gudorf said she hopes other researchers are inspired by the Indiana team’s successful proof-of-concept, and help move the treatment forward to—and through—clinical trials.
“We hope our approach leads to the birth of new therapeutics, which, in the hands of emergency personnel, could save even more lives,” said Bosquez-Berger.